 
In this case, we show that addition of hydroxide ion to lead(II) ion initially gives a white precipitate of lead(II) hydroxide.Īddition of excess hydroxide ion results in the formation of the colorless soluble tetrahydroxolead(II) complex ion.Ī characteristic test for lead(II) ion is the production of bright yellow lead(II) iodide:Īddition of excess iodide ion results in the formation of the colorless soluble tetraiodoplumbate(II) complex ion. Reaction of lead(II) ion with hydroxide ion The production of the gas can be illustrated by damp blue litmus paper turning red. The process continues until almost all of the hydrogen carbonate ion has been consumed.Īddition of any strong acid to a carbonate or hydrogen carbonate will give a release of carbon dioxide gas. Thus addition of calcium ion consumes the carbonate ion and the equilibrium shifts right to replenish the carbonate ion. HCO3-(aq) + HCO3-(aq) ? CO32-(aq) + H2O(l) + CO2(aq)Ĭalcium ion (small, high charge) forms insoluble calcium carbonate (high lattice energy as carbonate is high charge also). The hydrogen carbonate ion is in equilibrium with the carbonate ion in a disproportionation reaction: The indicator changes colour to indicate an acid.ĭisproportionation of the hydrogen carbonate ion Therefore oxidation number of Pb in PbSO 4 is +2.Weller, Overton, Rourke & Armstrong: Inorganic Chemistry 6e Chapter 14: Videos of chemical reactionsĪ lump of solid carbon dioxide is placed in water containing a few drops of universal indicator. To neutral that -2 charge, +2 charge should be there and it is given by Pb 2+ ion. PbSO 4 salt does not have an overall charge. what is the oxidation number of pb in pbso4 You know lead (Pb) is a metal and always form positive oxidation number and +2 is common in most compounds. Soluble chlorides, such as hydrochloric acid, precipitate white lead chloride. Sulfide ion is oxidized and hydrogen peroxide is reduced. Precipitate:Cl- (white), Br- (pale yellow), I- (yellow) No Precipitate:SO42-, PO43-,C2O42-, NO3- Add 6M NH3Add 1M BaCl2Add 0. With HCl, PbS is converted to PbCl 2 white precipitate.ĭissolves and produce colourless 2- aqueous solution. PbS is in solid state and H 2O 2 is in the aqueous state. H 2O 2 can be decomposed to H 2O and O 2. Two reactants, you cannot observe a white precipitate forming. You are provided PbS and aqueous H 2O 2 solution to produce PbSO 4, white precipitate. To take back white colour, PbS is treated with Lead carbonate reacts with H 2S forming black PbS. You can learn how a redox reaction is balanced in next few minutes. 
We have to follow some rules (steps) to balance a redox reaction. Balancing the reaction of PbS + H 2 O 2 PbSO 4 + H 2 O. When air is polluted with hydrogen sulfide (H 2S), PbS is a black precipitate and PbSO 4 is a white precipitate. Lead carbonate (PbCO 3) is used as a white pigment. Then simplified the stoichiometric coefficient.īalanced Equation PbS + 4H 2O 2 = PbSO 4 + 4H 2O See the following figure.Įxchange oxidation numbers. So difference is 6.Ĩ:6 ration is simplified as 4:3. Six oxygen atoms in H 2O 2 is at -1 are reduced to -2. 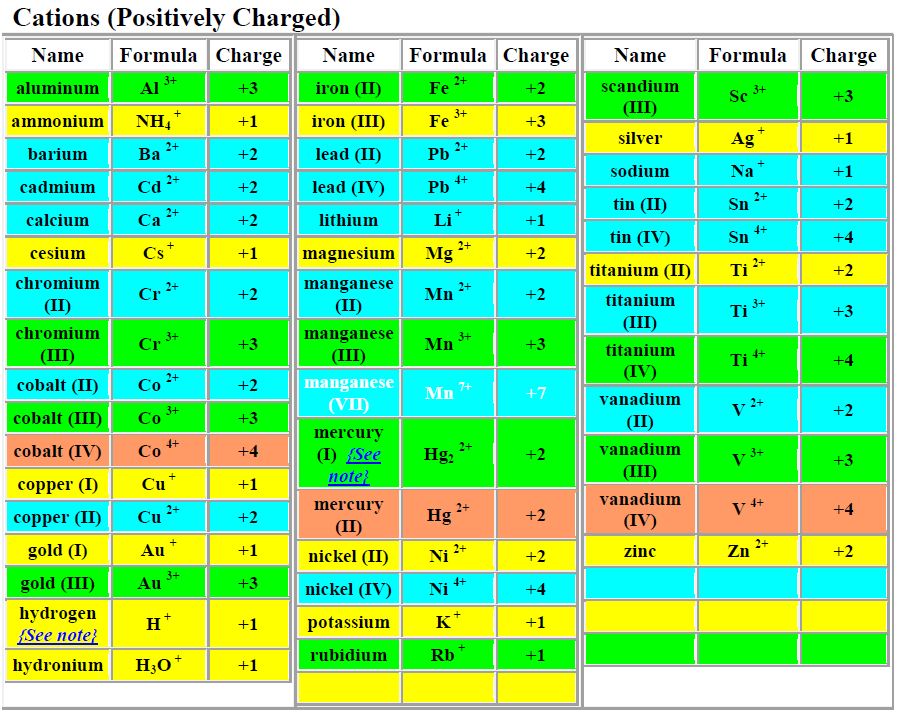
Sulfur atom at -2 oxidation state is oxidized to +6 oxidation state. Two balance oxygen atoms, we make three H 2O 2 in the left and two H 2O in the But number of oxygen atoms (reducing) is different. Number of sulfur atoms in both sides of the reaction is same (one). Cyclo alkanes Q.85 Consider equation: Pb+2 (aq) + 2OH (aq) precipitate of lead (II) hydroxide will be ofA. 
5 Upon heating to decomposition temperatures, it forms lead oxide (PbO) in the yellow form, litharge. Equal number of oxidizing and reducing atoms This cation hydrolyzes and, under progressively increasing alkaline conditions, forms Pb (OH) +, Pb (OH) 2 (aqueous), Pb (OH) 3, and other species, including several polynuclear species, e.g., Pb 4 (OH) 44+, Pb 3 (OH) 42+, Pb 6 O (OH) 64+. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
